8th Grade
Date: October 8-12,2018
Topic: Density
Home Learning: Study vocabulary terms and Density notes
Density Vocabulary Definitions
- 1. Mass - A measure of how much matter is in an object. (M=V x D)2. Weight- The amount of force the acceleration of gravity exerts on an object.3. Gravity- The force that attracts a body toward the center of the earth, or toward any other physical body having mass.4. Matter- Anything that has mass and takes up space.5. Density- The amount of matter in a given volume. (D=M/V)6. Volume- The amount of space an object takes up. (V= M/D)7. Displacement- The volume or weight of a fluid displaced by a floating body of equal weight8. Ratio- A way of concisely showing the relationship between two quantities of something. The quantitative relation between two amounts showing the number of times one value contains or is contained within the other9. Proportional- Relationship between two quantities is one in which the two quantities vary directly with one and other. If one item is doubled, the other, related item is also doubled.
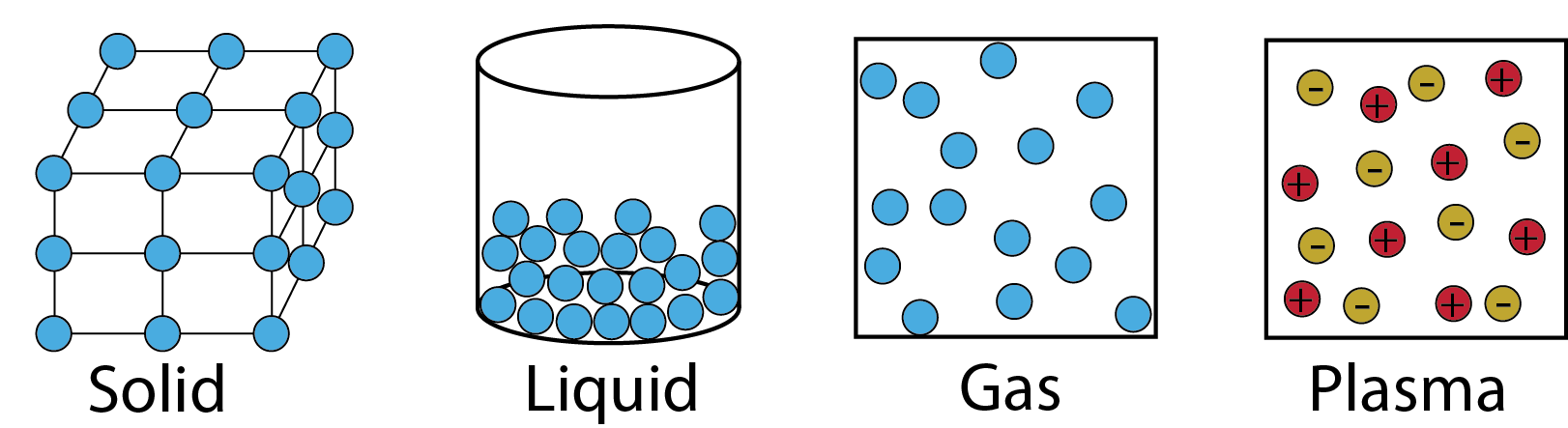
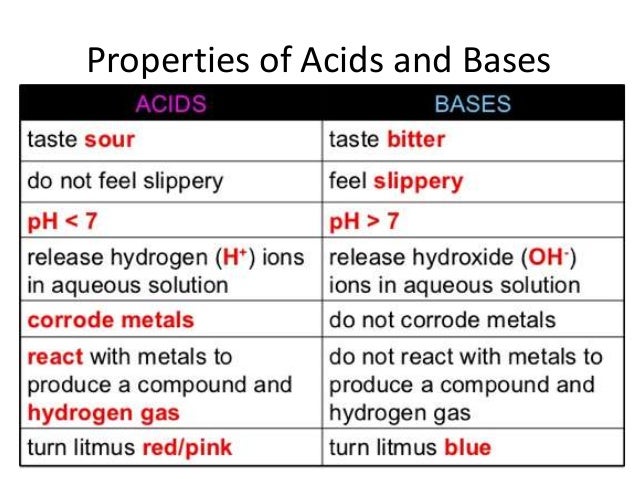
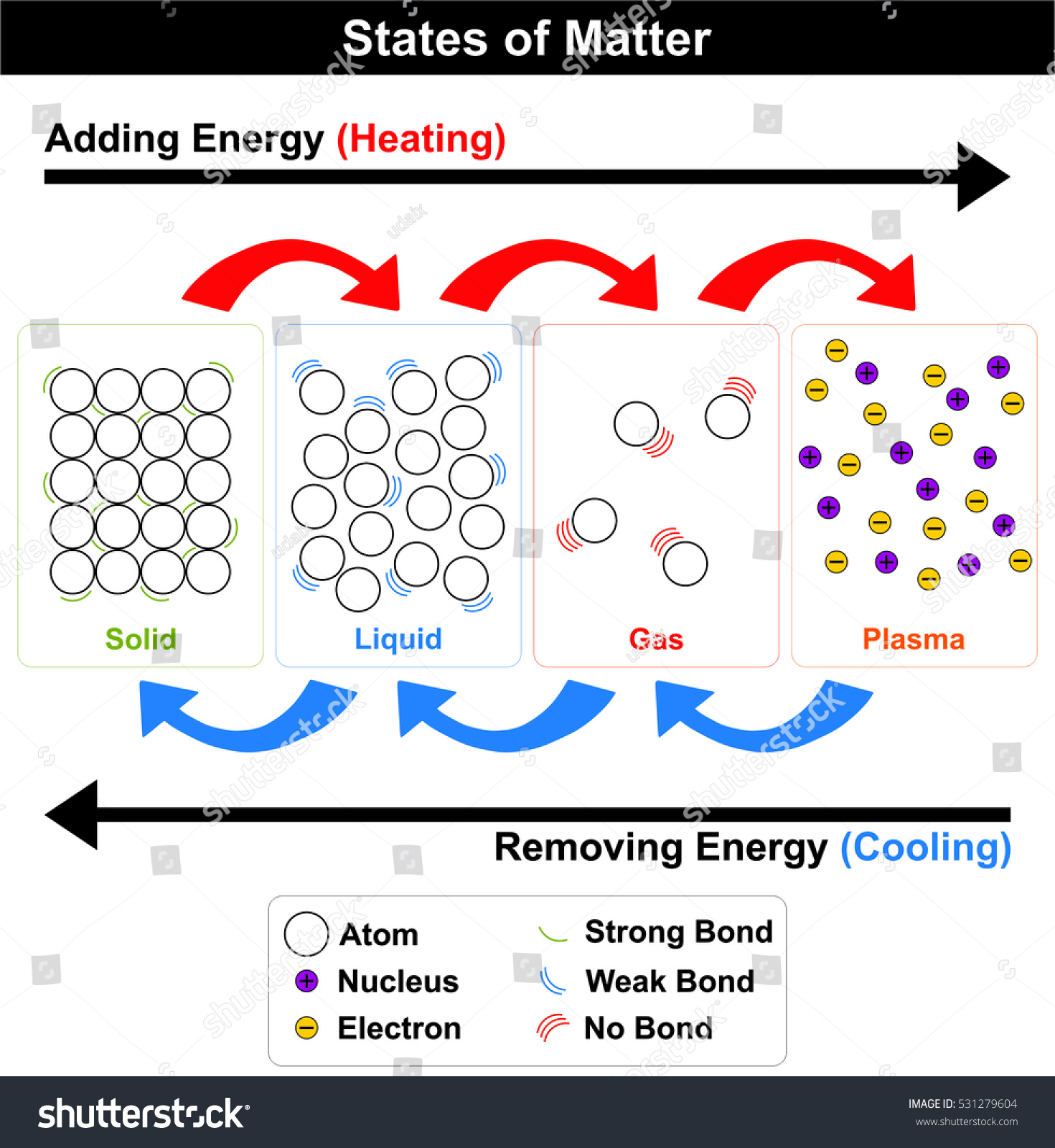
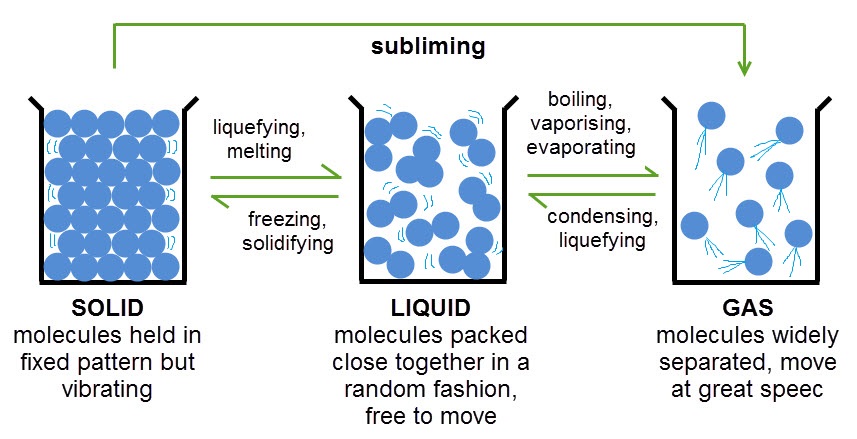
Anchor Chart 1
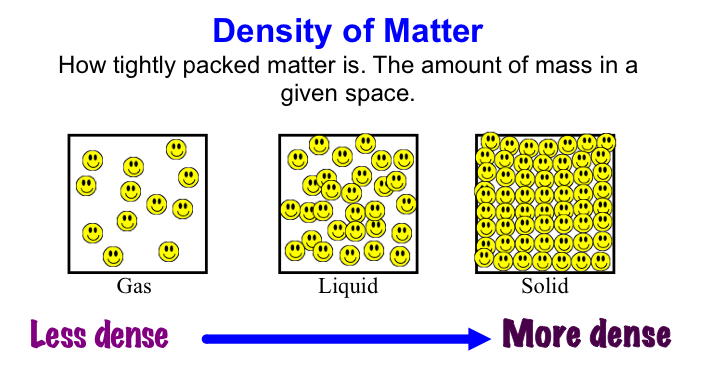
Anchor Chart 2
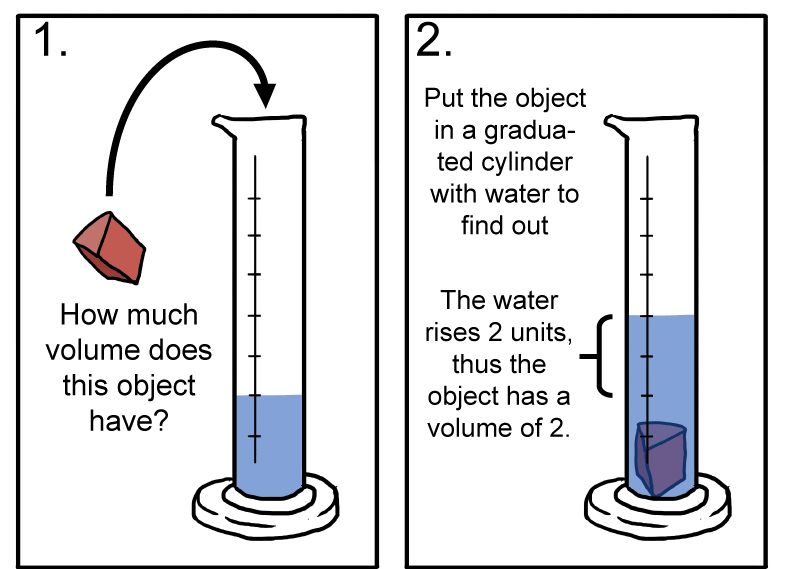
Anchor Chart 3
Station 1
Students will watch and create Cornell notes from a Density Tutorial (Click Density Tutorial to bring up the link). Notes are due at the beginning of class on 10/9/2018, notes will not be accepted late.